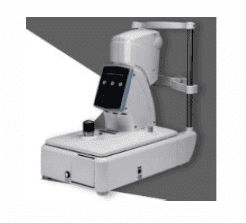
Tonometer - Non Portable
Order Code: 24258404.4
Category: General Lab Equipment V
Product Certification and Documentation: Valid Product Certificate from SRA Region: Required SRA Country of Origin: Specify Valid EFDA Product Certificate: Required Priority: 2 Product Information: GM...
SPECIFICATION
Product Certification and Documentation:
-
Valid Product Certificate from SRA Region: Required
-
SRA Country of Origin: Specify
-
Valid EFDA Product Certificate: Required
-
Priority: 2
Product Information:
-
GMDN/UMDN Code/Name: Specify
-
Clinical Purpose/Description:
Used for measuring intraocular pressure. -
Technical Specifications:
-
Pressure Range: 7-60 mmHg with 1 mmHg increments
-
Type: Non-contact tonometer
-
Chin Rest: Adjustable knob
-
Display: LED/LCD digital display screen
-
Measuring Unit: Vertical/horizontal and anterior/posterior movement
-
Adjustment Mechanism: Built-in joystick for adjustment
-
Accuracy: ±1 mmHg
-
System Configuration, Accessories, Consumables, and Other Components:
-
2 rolls of printer paper
-
2 fuses
-
1 dust cover
-
1 measuring nozzle cap
-
1 set of paper covers for chin rest
-
1 chin rest cover pin
-
1 cotton swab
-
1 calibration manual
-
1 spare LED/LCD display
Operating Environment:
-
Operating Temperature: +10°C to +32°C
-
Relative Humidity: <85%
Utility Requirements:
-
Power Supply: 220V, ±10%, 50 Hz
Standards and Safety Requirements:
-
Must comply with IEC 60601 (or equivalent) General Requirements for Electrical Safety
-
Valid Certificate from SRA Region countries required










 91-9829132777
91-9829132777